Introduction to Chemiluminescence Immunoassay (CLIA) and its Principle -part 2
Classification of Chemiluminescence Immunoassay Methods
Based on the different chemiluminescent agents in the chemiluminescent system, the chemiluminescence immunoassay (CLIA) method can be roughly divided into three categories: direct chemiluminescence immunoassay, chemiluminescent enzyme immunoassay, and electrochemiluminescence immunoassay.
3.1 Direct Chemiluminescence Immunoassay
Direct chemiluminescence immunoassay is a novel detection method that uses chemiluminescent labels for antibodies or antigens. Commonly used chemiluminescent labels include luminol and acridinium esters. Luminol labels should not be directly labeled and require the use of certain compounds to directly determine luminol through enhancement or inhibition. Direct chemiluminescence immunoassay has the following advantages: low background luminescence, fewer interfering factors in luminescent reactions, easy binding with proteins, constant photon yield, and stable labeling. However, the sensitivity of this method is slightly lower than enzyme-catalyzed reactions, and the luminescence duration is shorter. Therefore, competition methods are often used for the detection of small-molecule antigens, while sandwich methods are employed for large-molecule antigens, which are widely used in immunodiagnosis.
3.2 Chemiluminescent Enzyme Immunoassay
Chemiluminescent enzyme immunoassay (CLEIA) is a detection method that utilizes the interaction between an enzyme labeled on an antigen or antibody and a chemiluminescent substrate to produce a photon signal. Horseradish peroxidase (HRP) and alkaline phosphatase (AP) are the two most common enzyme labels in CLEIA. They have corresponding chemiluminescent substrates, with luminol commonly chosen for HRP and AMPPD for AP. The advantages of CLEIA include high sensitivity, stable binding of enzyme-labeled antigens or antibodies, long-lasting luminescence, easy recording and measurement. However, it has the drawbacks of slow reaction speed, low surface area, and small enzyme capacity. With the application of nanomaterials and catalysts, the surface volume of the solid-phase carrier has been expanded, increasing the binding capacity of proteins and enzymes, achieving signal amplification, and improving the sensitivity and detection efficiency of CLEIA.
3.3 Electrochemiluminescence Immunoassay
Electrochemiluminescence immunoassay (ECLIA) is a method for detecting and analyzing different proteins and biomolecules based on the principle of light emission in a chemical environment. It uses the chemiluminescent agent tris(2,2'-bipyridyl)ruthenium(II) [Ru(bpy)2+3] labeled on an antigen or antibody, and tripropylamine (TPA) as an electron donor. Through the electron transfer in the electric field, it generates chemiluminescence for detection. ECLIA has many advantages. Compared to chemiluminescence (CL), ECLIA is more selective, allowing for selective control of the generation of excited states by changing the electrode potential. Compared to photoluminescence (PL), ECLIA does not require a light source, effectively reducing issues such as light scattering and luminescent impurities. Compared to electrochemical methods, ECLIA has higher selectivity and fewer electrode fouling issues. However, in clinical testing, interference from electrochemically active materials and factors such as adsorbed protein membrane contamination can affect the results. ECLIA is particularly suitable for the detection of small-volume samples, is less prone to interference from hemolysis or turbid precipitates in the sample, and is easily automated.
Related Immunoassays
- Cardiac Markers
-
Tumor Marker
-
PGII
-
G17
- CA50
-
CA125
- CA242
-
CA15-3
- CA19-9
- CA72-4
-
Pepsinogens I (PGI)
-
Human Epididymis 4 (HE4)
- Prostate-Specific Antigen (PSA)
- Squamous Cell Carcinoma (SCC)
- Neuron-Specific Enolase (NSE)
- Cytokeratin 19 Fragment (CYFRA21-1)
- Human Progastrin-releasing Peptide (ProGRP Tumor Marker)
- Protein Induced by Vitamin K Absence or Antagonist-II (PIVKA II Tumor Marker)
- Alpha-fetoprotein(AFP)
-
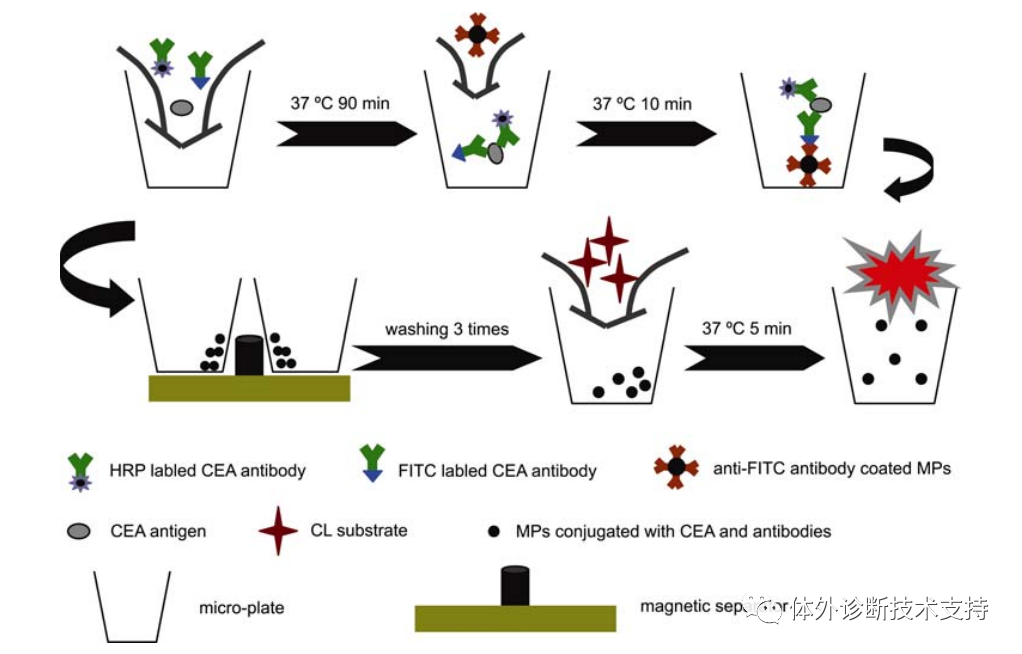
CEA
-
Human Chitinase 3-like 1
-
PGII
- Inflammatory Marker
- Infectious Disease
- Hormones
- Thyroid Function
- Glucose Metabolism
- Bone Marker
- Others
-
Heterophilic Blocking Reagent
- Animal Diagnostics