POCT Basic Technology Principle
01 Dry Chemistry Analysis Technology
Dry chemistry technology is compared to traditional wet chemistry technology, which uses the liquid in the tested sample as the reaction medium. The measured components directly react with the dry reagents fixed on the carrier, and are qualitatively observed by the naked eye or instrument detection (semi-quantitative) for chemical analysis.
Single-layer test paper technology:It includes single-item test paper and multi-item test paper. Single-item test paper can only test one item at a time, such as widely used blood glucose test paper, blood ammonia test paper, urine glucose test paper, etc. Multi-item test paper can simultaneously detect several, dozens, or even tens of items on one test strip, and its technology is relatively complex.
Multi-layer coating technology:It is to coat multiple reaction reagents in sequence on a film base to make a dry film. It is transplanted from photosensitive film production technology. Dry films made by multi-layer coating technology can be accurately quantified by instrument detection. For example, the dry plate chemistry analysis system currently used in clinical practice can be used for most blood chemistry components, such as proteins, carbohydrates, lipids, enzymes, electrolytes, non-protein nitrogen, and monitoring of some blood drug concentrations. The number of detectable items can reach dozens, which almost covers the commonly used clinical biochemical test items. Dry films made by multi-layer coating technology mainly include three layers: diffusion layer, reagent layer, and support layer.
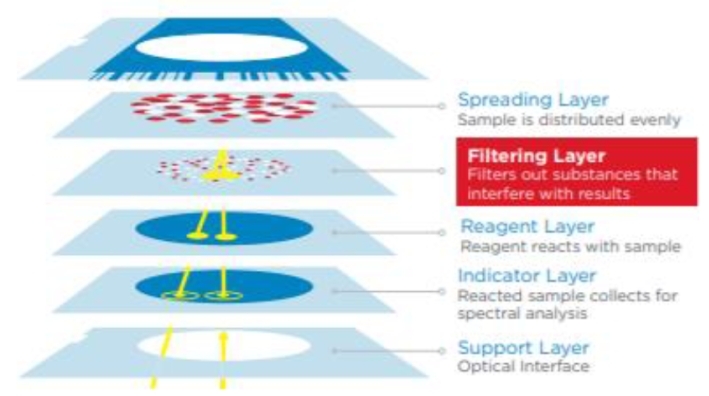
Principle of Dry Chemical Technology
02 Immunochromatography Technology
Immunochromatography assay (ICA) is a novel immunological analysis method that emerged in the early 1980s. It is a simple and rapid immunological detection method based on the principle of immunofiltration assay (IFA). The principle of immunochromatography is to first fix specific antibodies on a certain zone of nitrocellulose membrane. When the dried nitrocellulose is immersed in a sample (urine or serum), the sample will move forward along the membrane due to capillary action. When it reaches the area with fixed antibodies, the corresponding antigen in the sample will specifically bind to the antibody. If immunogold or immunoenzyme coloration is used, the zone will display a certain color. If immunogold or immunoenzyme staining is used, a colored band can be displayed, achieving specific immune diagnosis.
Colloidal gold is a stable colloid solution of negatively charged hydrophobic gold particles that can be aggregated into gold particles of a certain size under the action of reducing agents such as phosphorus, ascorbic acid, sodium citrate, and tannic acid. It is a stable colloidal state formed by electrostatic action. Immunogold labeling technology is similar to enzyme immunoassay, using colloidal gold as a label.
The spot immunofiltration assay was initially developed based on the spot ELISA and used enzyme-labeled conjugates, known as spot enzyme immunofiltration assay. In the early 1990s, spot immunofiltration assay using colloidal gold as a label (DIGFA), also known as colloidal gold immunodetection (CGID), was developed. In the CGID, there is no need for enzyme-substrate reaction, making it more convenient and faster, and it is increasingly widely used in clinical testing.
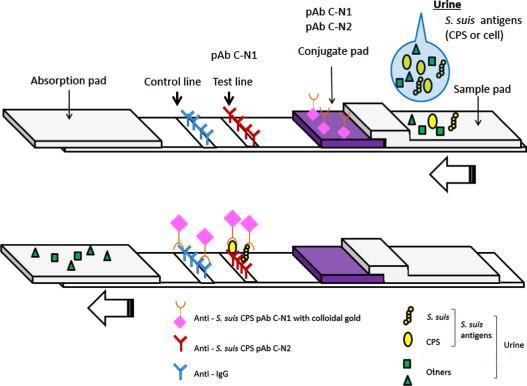
Principle of Immunochromatography Technology
03 Chemiluminescence Technology
Chemiluminescent immunometric assay (CLIA) is a non-radioactive immunoassay that has developed rapidly worldwide in the past decade. It is a highly sensitive trace analysis technology that has emerged after Enzyme-Linked Immunosorbent Assay (ELISA), Radioimmunoassay (RIA), and Fluorescence Immunoassay (FIR). Chemiluminescence Immunoassay combines highly sensitive chemiluminescence measurement technology with highly specific immune reactions, and is used for the detection and analysis of various antigens, semiantigens, antibodies, hormones, enzymes, fatty acids, vitamins, and drugs.
The direct labeling of antibodies or antigens with chemiluminescent agents in immunoassays is called chemiluminescence immunoassay. The commonly used direct chemiluminescent labeling agents include acridinium ester chemiluminescent agents. When acridinium ester is directly labeled with antibodies and then immune-reacted with the corresponding antigen in the sample, a solid-phase sandwich complex of antibody-captured antigen-labeled antibody is formed. At this time, adding an oxidant (H2O2) and NaOH to make an alkaline environment, acridinium ester can decompose and emit light without the need for a catalyst. The light emitted is received and recorded by a photomultiplier tube connected to a light collector, and the amount of the antigen in the sample is determined by calculating the integration of the emitted light, which is proportional to the amount of the antigen based on the standard curve.
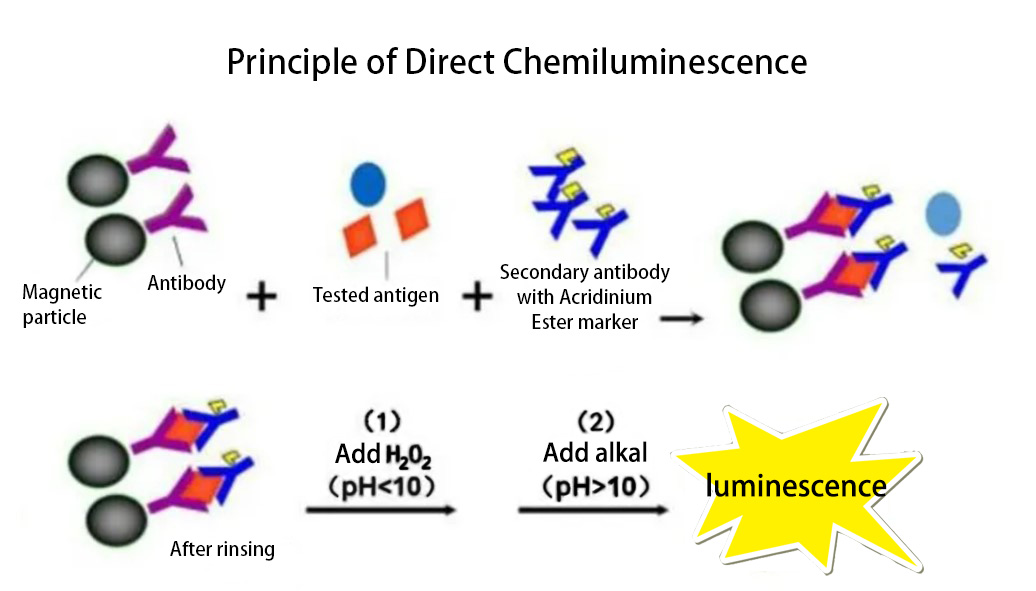
Enzyme-Linked Chemiluminescence Immunoassay (CLEIA) is an immunoassay that uses enzyme-labeled antigens or antibodies to perform immune reactions. The enzyme on the immune complex acts on the luminescent substrate, and luminescence occurs under the action of the signal reagent. The intensity of chemiluminescence is determined by the concentration of the enzyme. Horseradish peroxidase (HRP) and alkaline phosphatase (ALP) are commonly used labeling enzymes in chemiluminescence enzyme immunoassay, and luminescent substrates represented by luminol and AMPPD are used.
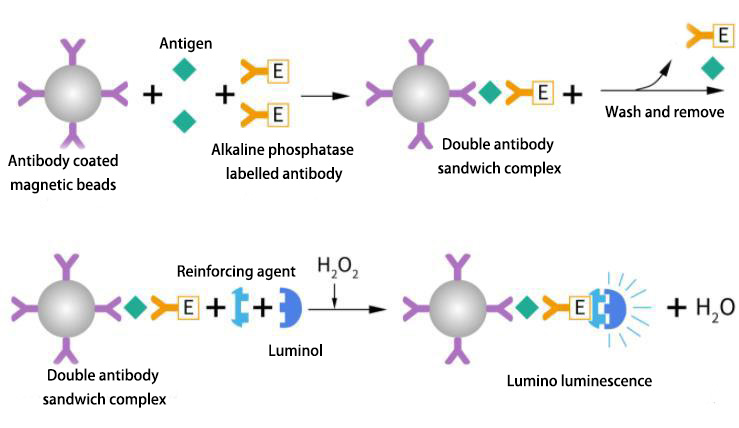
Enzyme-catalyzed chemiluminescence system based on luminol+HRP Electrochemiluminescence (ECLI) is a process in which a chemical reaction occurs between the electrochemical reaction product on the electrode or the reaction product of the electrode and a component in the solution when a certain voltage or current is applied to the electrode, and energy is released when the excited state transitions back to the ground state. Electrochemiluminescence Immunoassay (ECLIA) is an immunoassay that uses tris(2,2'-bipyridyl)ruthenium(II) as the electrochemiluminescent label of the antibody and tripropylamine (TPA) as the electron donor to produce a specific chemiluminescent reaction due to electron transfer in the electric field. It includes both electrochemical and chemiluminescent processes.
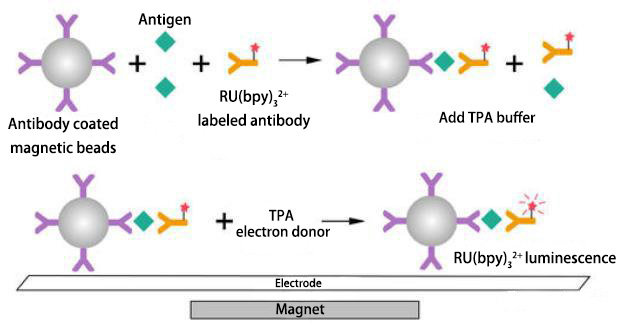
Electrochemiluminescence Principle
04 Biosensor Technology
Biosensors are devices that utilize the molecular recognition capabilities of active substances such as proteins, enzymes, and nucleic acids to convert the microscopic biological processes such as conformational changes and concentration changes of the detected substance into quantifiable and visible physical and chemical signals such as electrical signals and fluorescence signals, thereby achieving the purpose of detecting molecules such as proteins and nucleic acids.
Generally, biosensors consist of two parts. The first part is the biological or biochemical recognition element (or receptor), which is composed of sensitive materials that have the ability to recognize biological or chemical molecules, such as chemical sensitive membranes composed of electroactive substances and semiconductor materials, and biological sensitive membranes formed by enzymes, microorganisms, DNA, etc. The second part is the signal transducer, which is mainly composed of electrochemical or optical detection elements such as current and potential measuring electrodes, ion-sensitive field-effect transistors, piezoelectric crystals, etc.
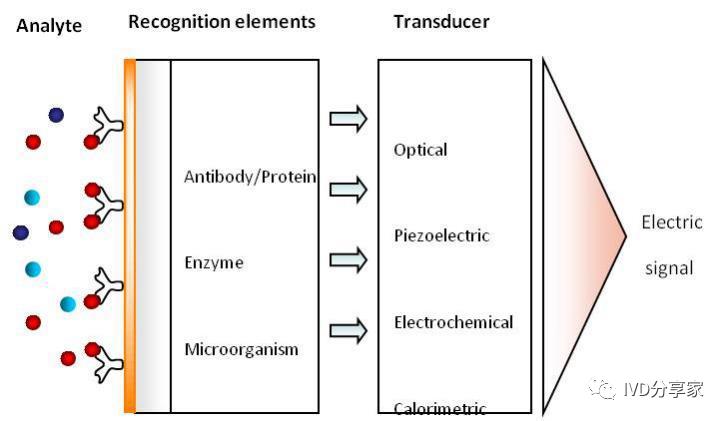
Diagram of biosensor
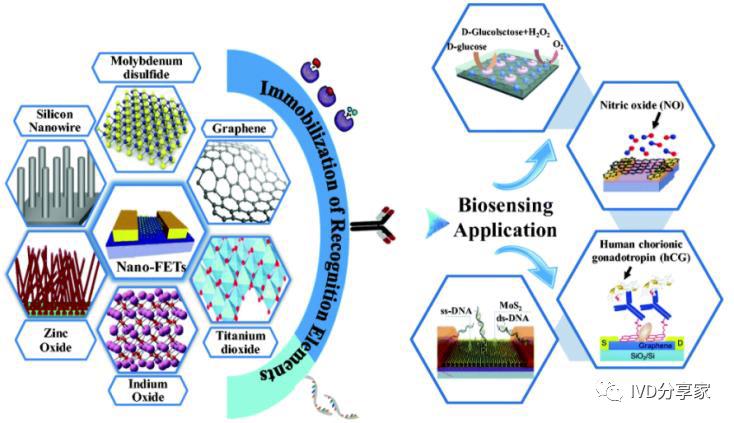
Materials and Applications of Biosensor
05 Biochip Technology
Biochip technology refers to the technology that immobilizes a large number of probe molecules on a support and hybridizes with fluorescently labeled DNA or other sample molecules (such as proteins, factors, or small molecules) to obtain the quantity and sequence information of sample molecules by detecting the hybridization signal intensity of each probe molecule. Since commonly used glass/silicon wafers are used as solid supports, and the preparation process simulates the manufacturing technology of computer chips, it is called biochip technology.
Currently, biochips can be divided into gene chips (or DNA chips), protein chips, cell chips, and lab-on-a-chip (LOC). They have the advantages of high sensitivity, short analysis time, and simultaneous analysis of multiple items. They integrate many analytical steps involved in life science research using microelectronics, micromechanics, physical technology, sensor technology, and computer technology to make the sample detection and analysis process continuous, integrated, and miniaturized. It can also promote the construction of micro-laboratories.
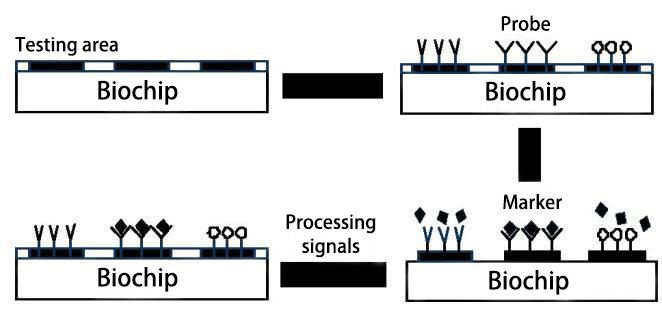
Principle of Biochip Technology
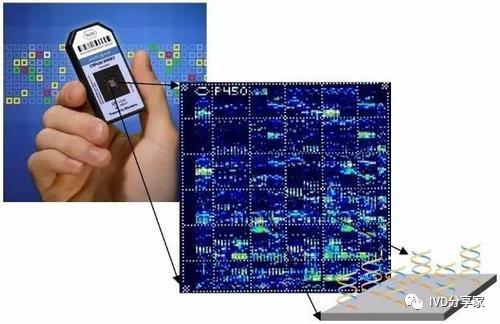
Typical Products of Gene Chips
06 Microfluidics technology
Microfluidics is a scientific technology characterized by precise control and manipulation of fluids at the micro and nanoscale. It integrates the basic operational units of medical analysis, such as sample preparation, reaction, separation, and detection, into a chip of a few square centimeters. The process is automated, allowing for accurate control of the quantity and flow rate of samples and reagents. The main feature and greatest advantage of microfluidics is the flexible combination and scale integration of various unit technologies on a controllable micro-platform.
Implementation methods of microfluidics include pneumatic pushing, centrifugal pushing, droplet microfluidics, digital microfluidics, paper-based microfluidics, etc. Currently, the most widely used methods are the first two.
Pneumatic pushing microfluidics mainly uses air pressure to drive the movement of fluids in the chip and is the most common in the industrialization of microfluidics, such as BioMerieux's FilmArray, Roche Diagnostics' cobas Liat PCR System, AtlasGenetics' io System, BoHui Innovation's HPV molecular diagnostic automatic analyzer, and Huamai Xingwei's M2 miniaturized chemiluminescence analysis system. Centrifugal pushing microfluidics uses centrifugal force to drive chip movement and also plays an important role in the microfluidics industry, such as the Piccolo Xpress instant biochemical analyzer from Abaxis in the United States and the Pointcare M from Tianjin MNCHIP.
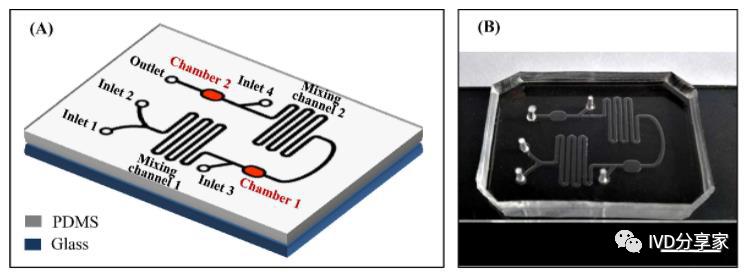
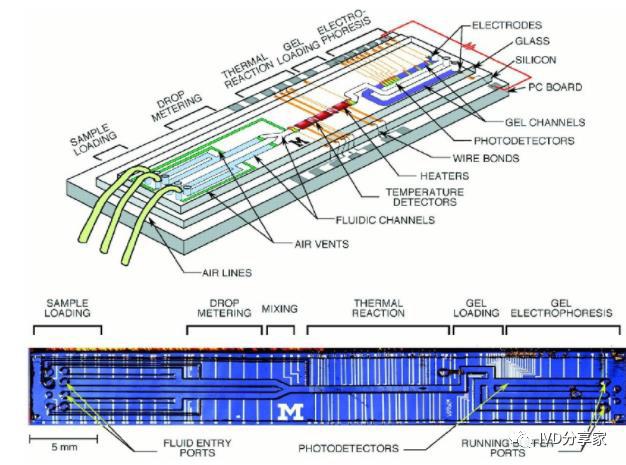
Microfluidics technology/ Microfluidic chip technology
07 Other Technologies
1、Infrared and far-infrared spectrophotometry technology
It is commonly used in the production of transdermal detection instruments and can be used to detect various components such as blood hemoglobin, bilirubin, and glucose. These detection instruments can continuously monitor the target components in a patient's blood without the need for blood sampling, thereby avoiding potential cross-infection and blood specimen contamination, reducing the cost of each test, and shortening the reporting time. However, the accuracy of such transdermal detection results needs to be improved.
2、Selective electrode technology
It is combined with biosensor and chemical sensor technology, has been used to produce portable instruments for rapid detection of blood gases (such as pH, PCO2, PO2, etc.) and electrolytes (such as K+, Na+, Cl-, etc.), and has been widely used in clinical settings.
3、Automatic identification technology
①Barcode recognition technology
To successfully implement barcodes, POCT requires the ability to identify all items that need to be identified by the system, including patients, operators, test strips, and quality control (QC) materials. Barcodes on test strips and QC materials simplify the process of entering key data into the system. Currently, the POCT industry has accepted barcodes, and many POCT devices have built-in barcode scanners to improve data entry.
②Radio frequency identification technology
RFID technology is a new technology that is still developing and is not subject to the limitations of barcodes. RFID systems use a small electronic tag composed of a small integrated circuit connected to a miniature antenna, which is read by an RFID scanner, similar to how a barcode reader reads printed barcodes. Data about the tagged object is stored in the memory of the integrated circuit, and the information is wirelessly transmitted from the RFID tag to the reader via radio frequency signals.
By fully utilizing RFID technology, the POCT process can be greatly simplified. POCT devices with built-in RFID readers can collect information from all objects with RFID tags within their reading range to quickly record each data element and create error-free, medically effective test results.